Vapor Pressure Vs Temperature Graph Apr 19 2021 nbsp 0183 32 The recent meaning of vapor from OED quot Vapor In modern scientific use a fluid that fills a space like a gas but being below its critical temperature can be liquefied by pressure alone quot This
Mar 23 2018 nbsp 0183 32 Vape 1980 Vaping To inhale water vapor 2021 3 16 vapor 3 3 0 3 2 0 3 2 0
Vapor Pressure Vs Temperature Graph

Vapor Pressure Vs Temperature Graph
https://i.ytimg.com/vi/JIokrlP2bZ8/maxresdefault.jpg
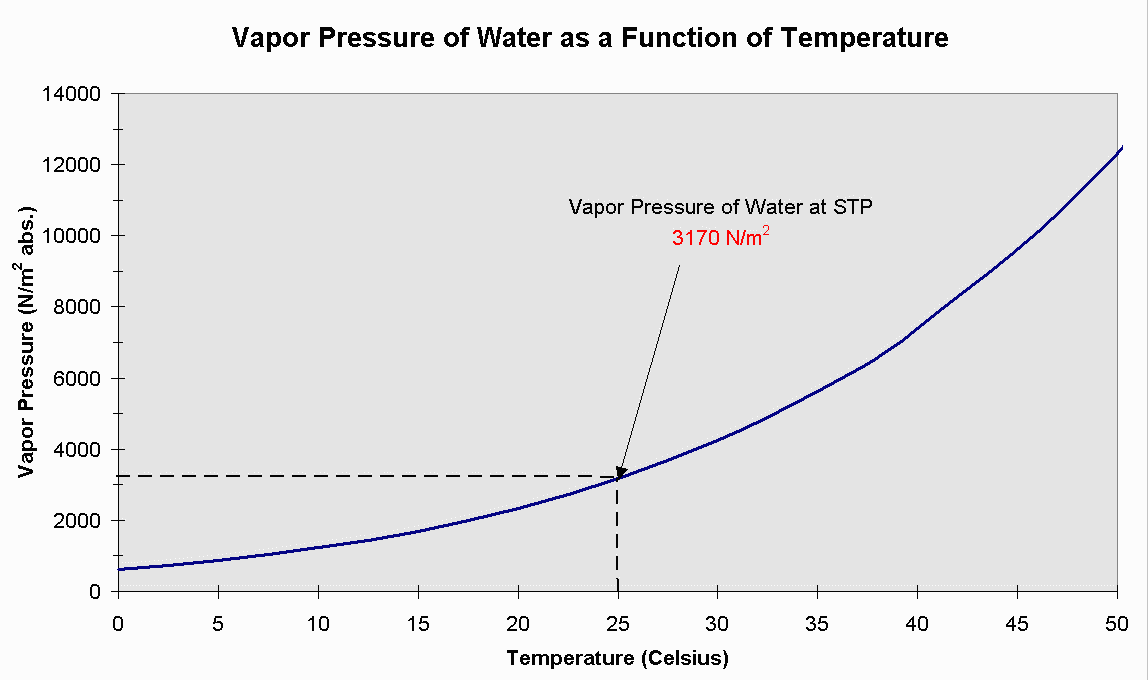
Vapor Pressure Vs Temperature
https://www.caee.utexas.edu/prof/kinnas/319lab/book/ch1/props/GIFS/vappres2.gif
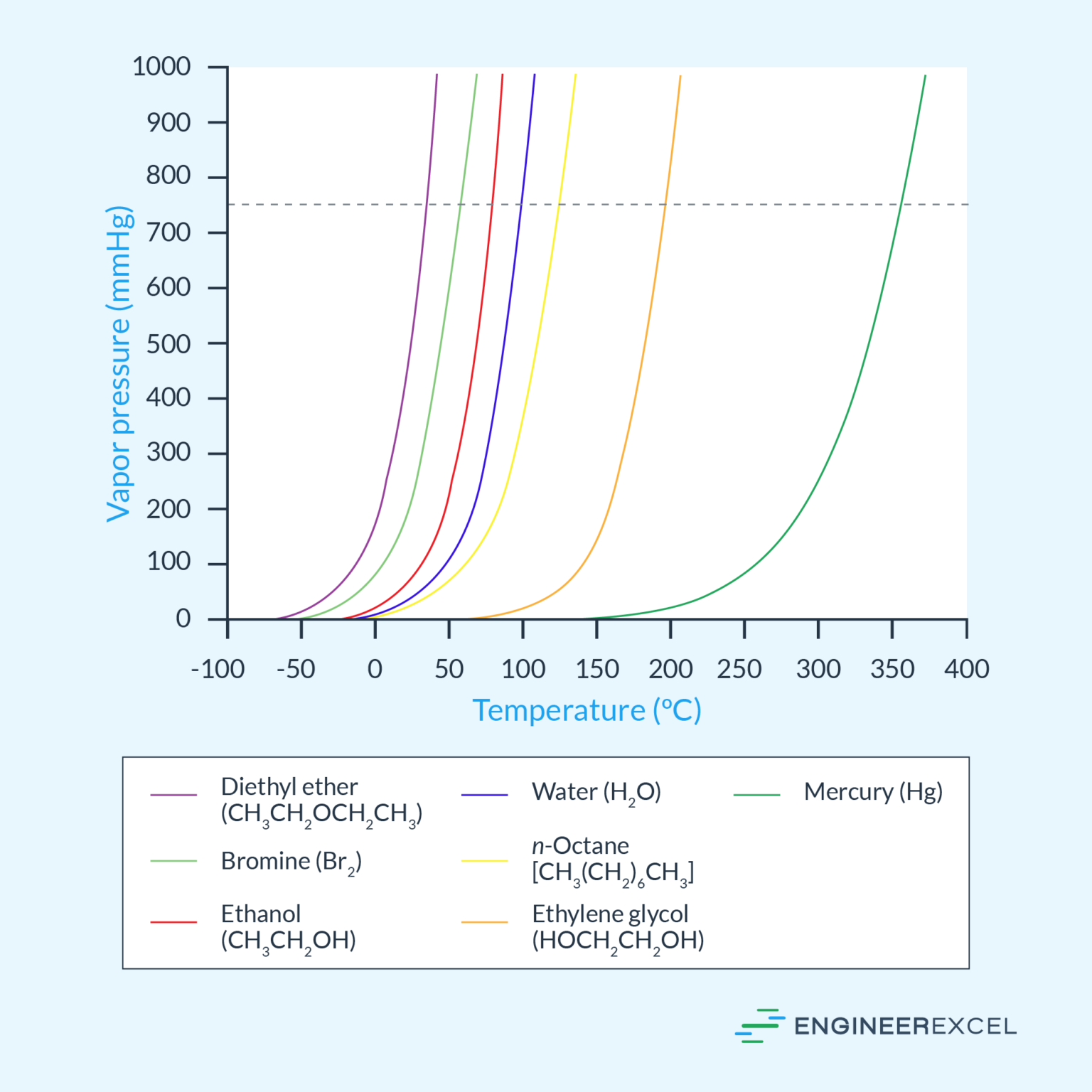
Vapor Pressure A Comprehensive Overview EngineerExcel
https://engineerexcel.com/wp-content/uploads/2023/12/vapor-pressure-vs-temperature-diagram-of-different-1536x1536.png
vue vue vapor 2024 10 24 Vapor leaf SEO leaf VaporLeaf
May 6 2024 nbsp 0183 32 At room temperature and 1 atm pressure liquid water should be a compressed liquid Now if that is the case then why do we have water vapor at these conditions I can t make sense of this Oct 2 2024 nbsp 0183 32 The equilibrium vapor pressure varies with temperature because of two factors 1 kinetic energy of particles and 2 number density Both increase non negligibly with temperature The
More picture related to Vapor Pressure Vs Temperature Graph
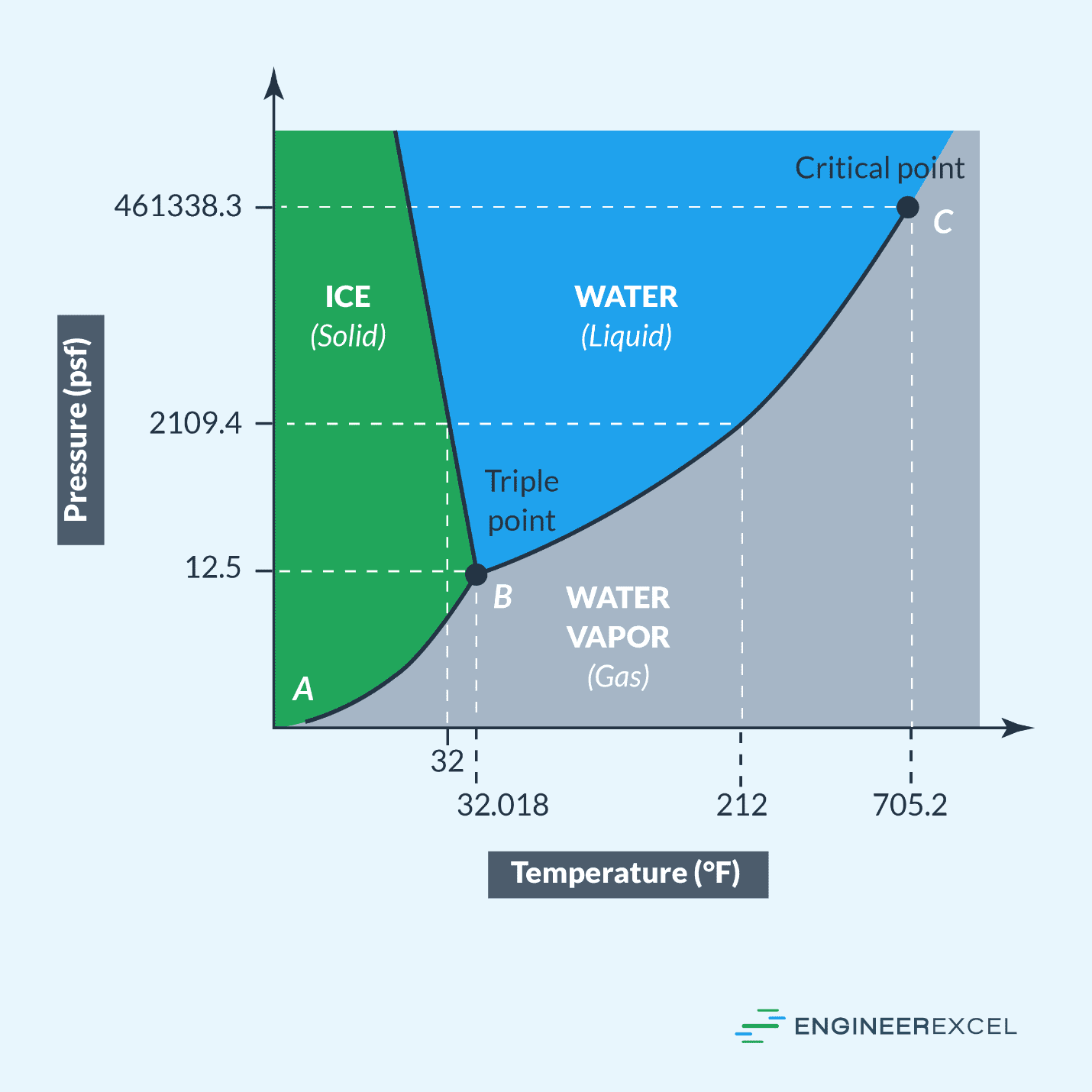
Pressure Temperature Graphs Explained EngineerExcel
https://engineerexcel.com/wp-content/uploads/2023/03/pressure-temperature-graph.webp
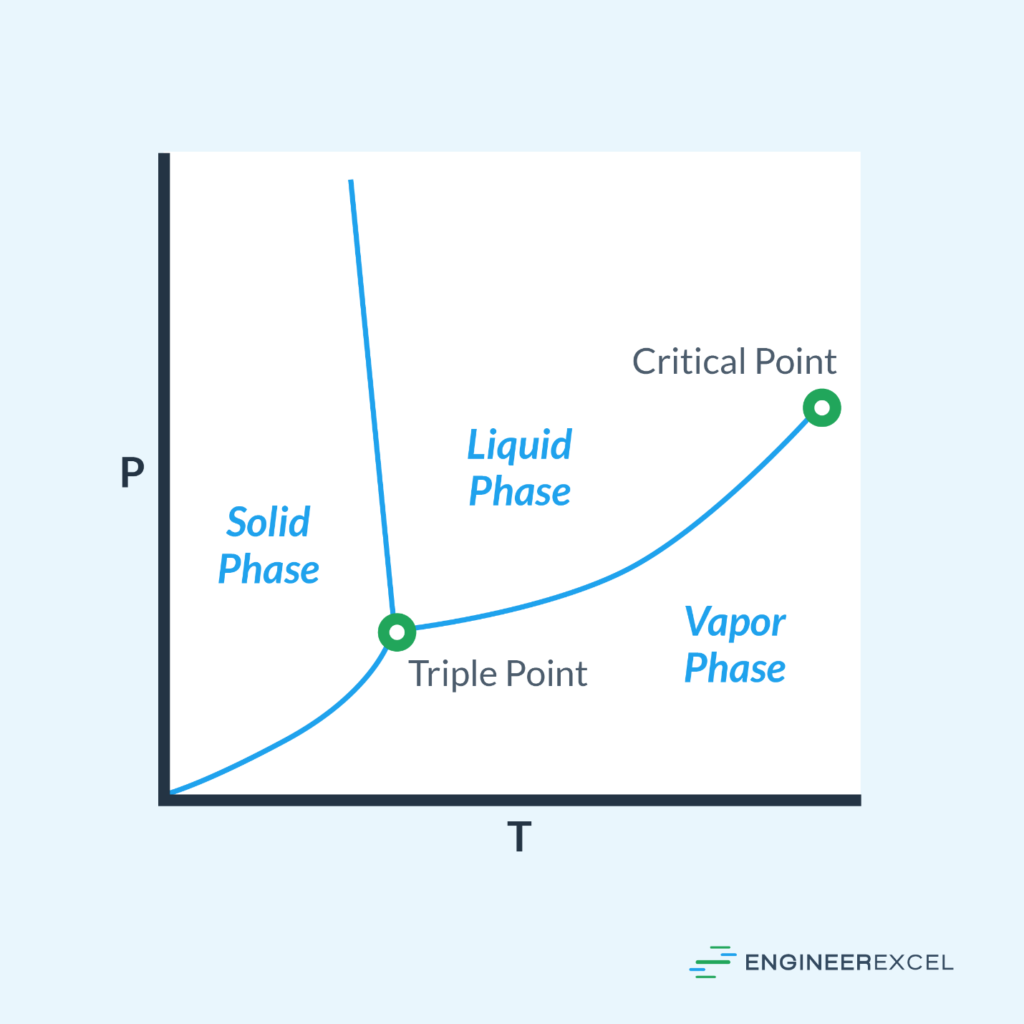
Pressure Temperature Graphs Explained EngineerExcel
https://engineerexcel.com/wp-content/uploads/2023/03/pressure-temperature-diagram-1024x1024.png
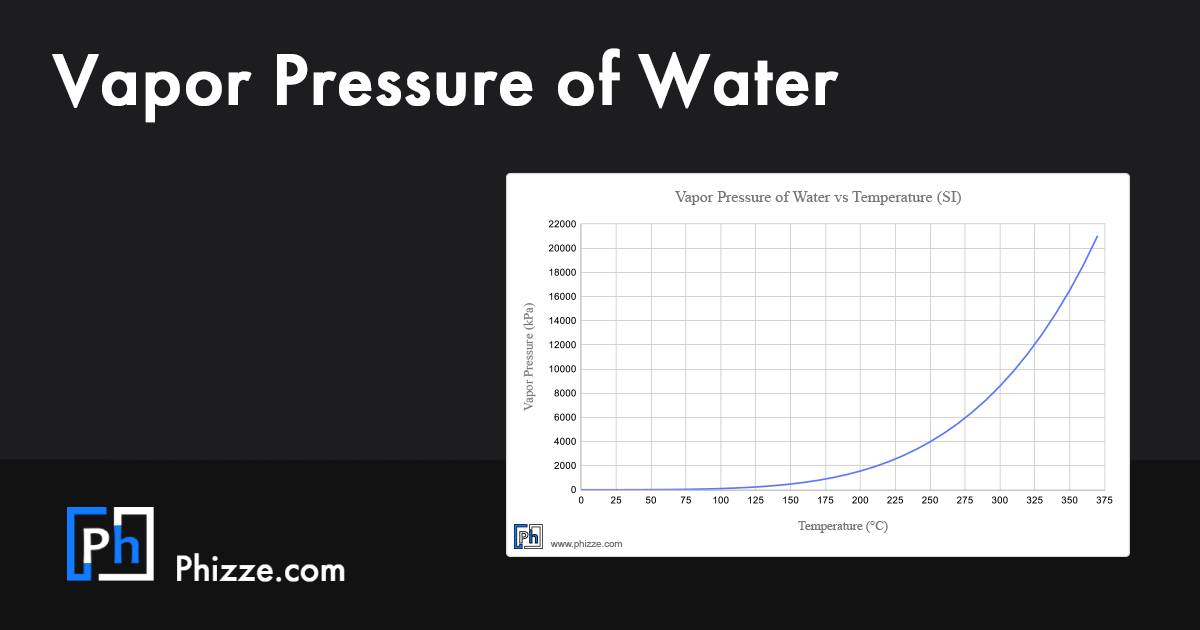
Vapor Pressure Of Water Vs Temperature Table
https://matmake.com/imgs/share/vapor-pressure-of-water-share.png
Jun 14 2023 nbsp 0183 32 I ve been reading about vapor pressure and it s a bit confusing For example when water is in equilibrium between the solid and liquid phase my textbook says that the vapor pressure is Jan 23 2014 nbsp 0183 32 Was looking at Henry s law and Raoult s law constants and there seemto be lots of equations involved Henry s law involves partial pressure and the latter involves the vapor pressure
[desc-10] [desc-11]
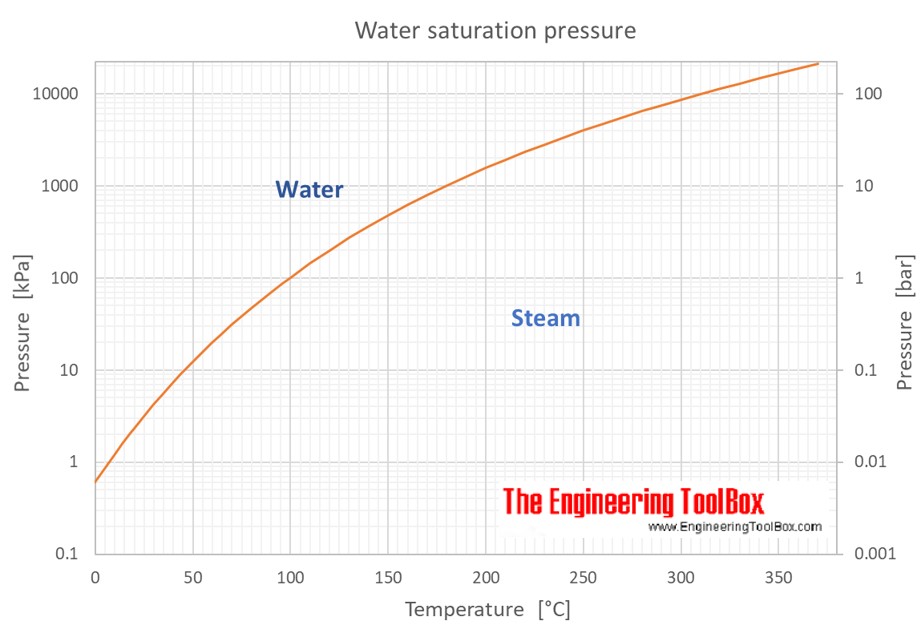
What Is Saturated Water Vapor Infoupdate
https://www.engineeringtoolbox.com/docs/documents/599/Water_saturation_pressure_C.jpg

What Is Saturated Water Vapor Infoupdate
https://www.researchgate.net/publication/225171795/figure/fig2/AS:349482043625504@1460334420724/Saturation-vapor-pressure-for-H-O-vs-temperature-The-average-amounts-of-water-vapor-that.png
Vapor Pressure Vs Temperature Graph - May 6 2024 nbsp 0183 32 At room temperature and 1 atm pressure liquid water should be a compressed liquid Now if that is the case then why do we have water vapor at these conditions I can t make sense of this