Solubility Curves Explained Solubility is the ability of a solute to dissolve in a solvent to form a solution This is the property that allows things like sugar molecules to dissolve in a cup of coffee
Solubility is the maximum concentration of a solute that can dissolve in a specific amount of a solvent at a given temperature The process through which a solute in its solid liquid or gaseous phase The solubility of a solute in a particular solvent is the maximum concentration that may be achieved under given conditions when the dissolution process is at equilibrium
Solubility Curves Explained

Solubility Curves Explained
https://i.ytimg.com/vi/D7M_Ey_VNmw/maxresdefault.jpg

Solubility Curves Explained YouTube
https://i.ytimg.com/vi/BDv_Y1LEZfU/maxresdefault.jpg?sqp=-oaymwEmCIAKENAF8quKqQMa8AEB-AH-CYAC0AWKAgwIABABGGUgZShlMA8=&rs=AOn4CLBjgaSK6k7e8j2DnGulhZ3itbZVJA

Solubility Curves And Practice Problems YouTube
https://i.ytimg.com/vi/xxy7o4I3BMQ/maxresdefault.jpg
Discover what solubility means factors affecting it and real life examples for easy student understanding Apr 3 2026 nbsp 0183 32 Solubility of one fluid liquid or gas in another may be complete totally miscible e g methanol and water or partial oil and water dissolve only slightly
Jun 9 2025 nbsp 0183 32 Solubility is defined as the maximum quantity of a substance that can be dissolved in another It is the maximum amount of solute that can be dissolved in a solvent at equilibrium which Aug 13 2025 nbsp 0183 32 Solubility is how well a solute dissolves in a solvent to form a solution including liquid in liquid solid in liquid and gas in liquid solutions
More picture related to Solubility Curves Explained

Solubility Curves And Key Terms YouTube
https://i.ytimg.com/vi/upTDJSLHQZ8/maxresdefault.jpg
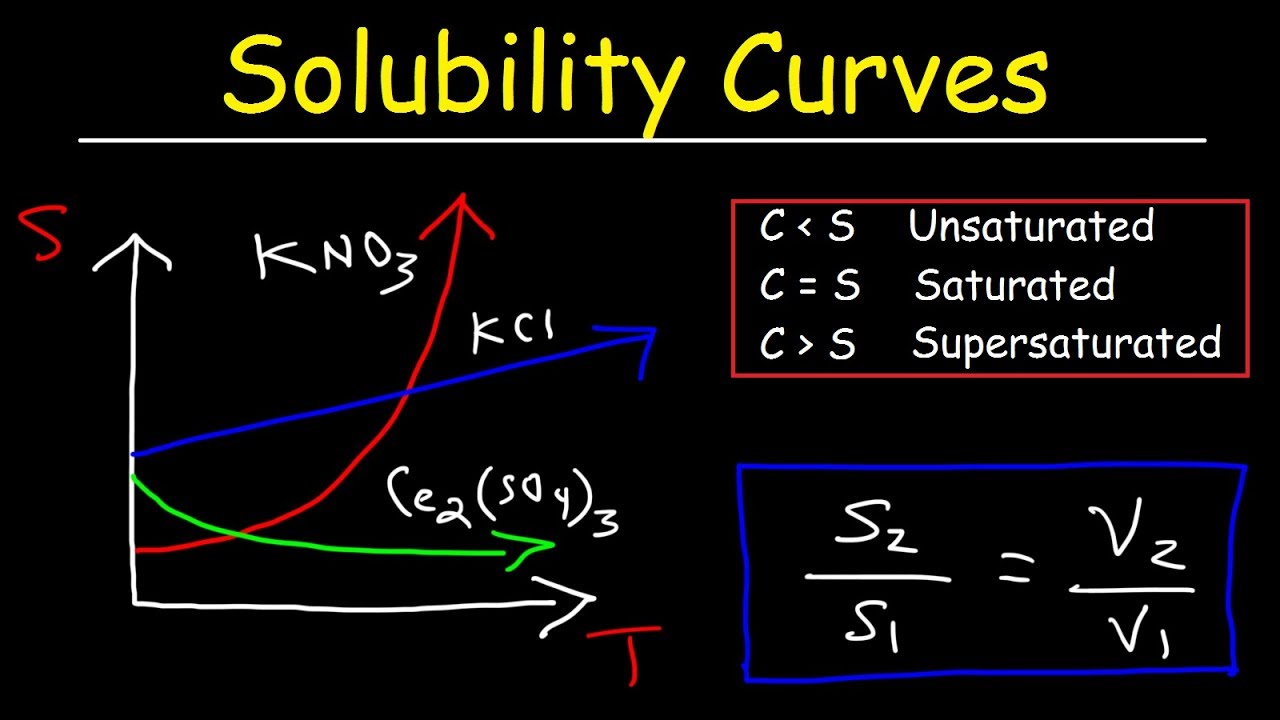
Solubility Curves Basic Introduction Chemistry Problems YouTube
https://i.ytimg.com/vi/VKmyj1z8T8U/maxresdefault.jpg

Solubility Curves 2 YouTube
https://i.ytimg.com/vi/RUURRhdLf8w/maxresdefault.jpg
There are a number of patterns in the data obtained from measuring the solubility of different salts These patterns form the basis for the rules outlined in the table below which can guide predictions of The solubility of a solute in a particular solvent is the maximum concentration that may be achieved under given conditions when the dissolution process is at equilibrium
[desc-10] [desc-11]
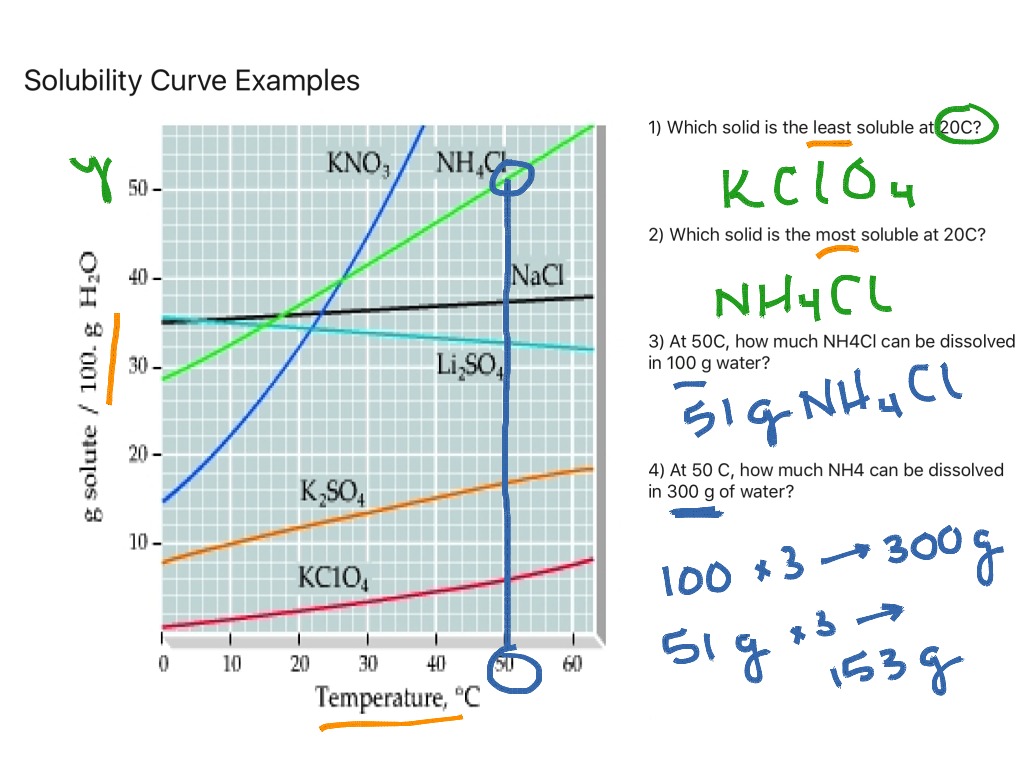
Solubility Solubility Curves Science Chemistry ShowMe
https://showme0-9071.kxcdn.com/files/101150/pictures/thumbs/2262464/last_thumb1453428064.jpg

Crystal16 Spincotech
https://spincotech.com/wp-content/uploads/2023/10/product-3-2.jpg
Solubility Curves Explained - Apr 3 2026 nbsp 0183 32 Solubility of one fluid liquid or gas in another may be complete totally miscible e g methanol and water or partial oil and water dissolve only slightly