Explain Empirical And Molecular Formula Oct 12 2020 nbsp 0183 32 The empirical formula of a substance can be calculated from its percent composition and the molecular formula can be determined from the empirical formula and the compound s molar mass The
There are two types of formulas empirical and molecular Empirical Formula Lowest whole number ratio of the elements in a compound Molecular Formula Actual whole number ratio of the elements in a compound The Molecular formulas are derived by comparing the compound s molecular or molar mass to its empirical formula mass As the name suggests an empirical formula mass is the sum of the average atomic masses of all the atoms
Explain Empirical And Molecular Formula

Explain Empirical And Molecular Formula
https://image1.slideserve.com/2694403/empirical-formula-vs-molecular-formula-n.jpg

Finding Empirical Formula Chemical Formula And Equation Chemistry
https://i.ytimg.com/vi/XUTAfia9UVY/maxresdefault.jpg

Calculating Empirical And Molecular Formula YouTube
https://i.ytimg.com/vi/cYvdlvwJ8tM/maxresdefault.jpg
The empirical formula of a compound is the simplest whole number ratio of atoms of each element in a compound The molecular formula shows the actual number of atoms of each Jan 22 2019 nbsp 0183 32 The empirical formula is the ratio of elements present in the compound The subscripts in the formula are the numbers of atoms leading to a whole number ratio between them Examples of Molecular and Empirical
In this explainer we will learn how to define determine and convert between a compound s empirical and molecular formulas The composition of a molecule can be expressed as a Aug 2 2022 nbsp 0183 32 The empirical formula of a compound is defined as the formula that shows the ratio of elements present in the compound but not the actual numbers of atoms found in the molecule The ratios are denoted by subscripts
More picture related to Explain Empirical And Molecular Formula

Molecular Formula
https://i.ytimg.com/vi/0Ikv_o7gcAE/maxresdefault.jpg

Difference Between Empirical And Molecular Formula Class 11 Chemistry
https://i.ytimg.com/vi/0yp20L7_XzU/maxresdefault.jpg
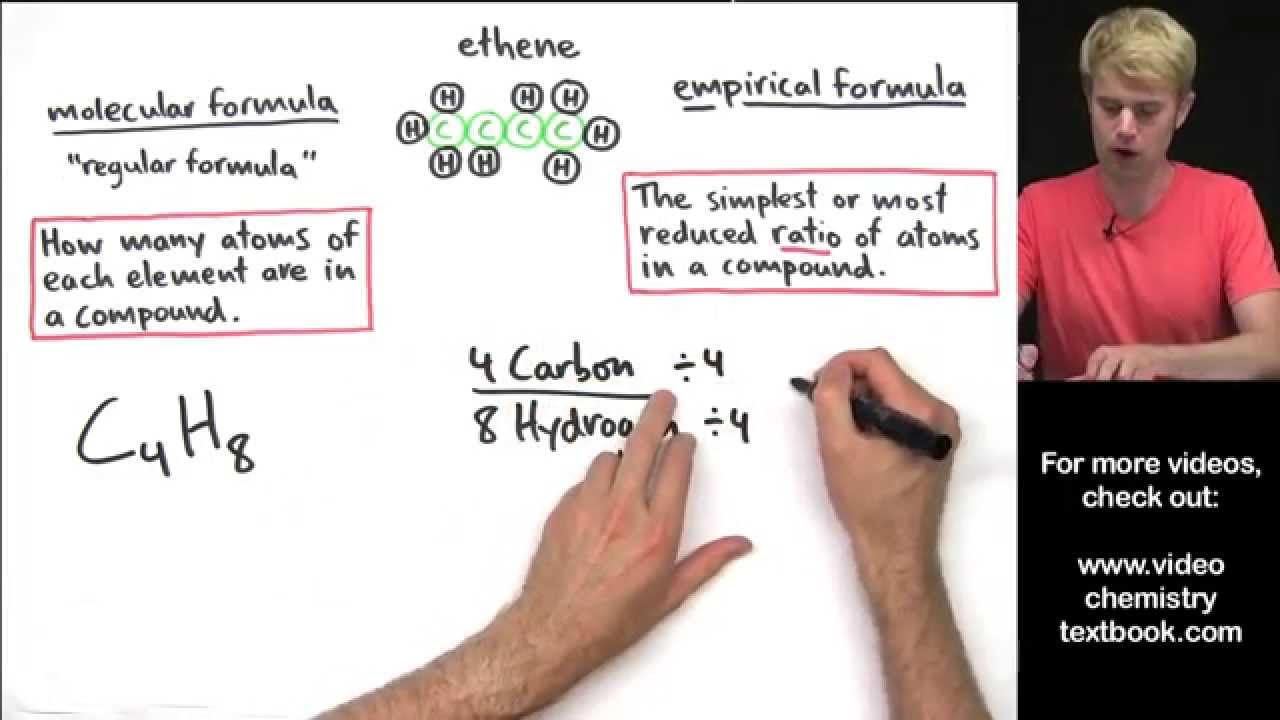
Empirical And Molecular Formulas Lessons Blendspace
https://i.ytimg.com/vi/wnRaBWvhYKY/maxresdefault.jpg
Feb 17 2020 nbsp 0183 32 The empirical formula of a chemical compound is a representation of the simplest whole number ratio between the elements comprising the compound The molecular formula is the representation of the Sep 4 2024 nbsp 0183 32 The empirical formula is the simplest whole number ratio of the atoms of each element present in one molecule or formula unit of the compound E g the empirical
Therefore the empirical formula can be defined as the simplest whole number ratio of the constituent atoms of a sample compound Let s learn more about the empirical and The empirical formula of a substance can be calculated from its percent composition and the molecular formula can be determined from the empirical formula and the compound s molar

Difference Between Empirical And Molecular Formula Infographic
https://i.pinimg.com/736x/86/f6/14/86f6149a4ab82bf14abda03bd40cd6d1.jpg

Molecular Vs Empirical Formula Slideshare
https://image1.slideserve.com/3095492/empirical-and-molecular-formula-notes-n.jpg
Explain Empirical And Molecular Formula - High school chemistry Course High school chemistry gt Unit 5 Lesson 2 Mole calculations Empirical molecular and structural formulas Worked example Calculating mass percent