Mole Ratio To Mass Ratio Mole can be defined as the amount of substance that contains the same number of entities atoms molecules ions etc as the number of atoms present in 12g of the carbon 12 isotope
Mole and Equivalent Weight If we ask you to count the total number of stars in the sky can you do that No Similarly scientists can not count the exact number of atoms and molecules as they are very tiny An amount of 1 mole of N 2 and 4 moles of H 2 are allowed to react in a vessel and after reaction water is added Aqueous solution required 1 mole of H C l for complete reaction
Mole Ratio To Mass Ratio

Mole Ratio To Mass Ratio
https://i.ytimg.com/vi/e1o2YDU7gcM/maxresdefault.jpg

Mole Conversion Worksheet And Activity
https://i.pinimg.com/originals/6c/33/0f/6c330fc3f0dafbf002c3707e72fc54ce.jpg

Stoichiometry Mole To Mole Conversions Molar Ratio Practice Problems
https://i.ytimg.com/vi/3zmeVamEsWI/maxresdefault.jpg
A magnesium ribbon when burnt in air left an ash containing M gO and M g3N 2 The ash was found to consume 0 6 mole of HCl when it was taken in solution according to the following reaction M g3N 2 One mole of an ideal monoatomic gas initially at 1200 K and 64 atm is expanded to a final state at 300 K and 1 atm To achieve the above change a reversible path is constructed that involves an adiabatic
The heat of neutralization of a strong base and a strong acid is 57 kJ The heat released when 0 5 mole of H N O3 solution is added to 0 20 moles of NaOH solution is The vapour density of a gaseous mixture containing only A r and N 2 O 4 gases is 40 When the mixture is left for some time the vapour density decreased and finally becomes 37 5 It happened due to
More picture related to Mole Ratio To Mass Ratio

Mole Ratios And Stoichiometry YouTube
https://i.ytimg.com/vi/fjRcgPIb2LI/maxresdefault.jpg

How To Calculate The Molar Mass Of A Compound Quick Easy YouTube
https://i.ytimg.com/vi/c_zHROisdP4/maxresdefault.jpg

Calculating Mass Percentage Mole Fraction Molarity And Molality Of
https://i.ytimg.com/vi/xkwI1cJOulw/maxresdefault.jpg
291 24 H Which statement is true for solution of 0 020 M H2SO4 DPMT 2136 a 2 litre of the solution contains 0 020 mole of SO2 fo 2 litre of the solution contains 0 080 mole of H30 C i litre of the P A 235y 125xy mm of H g P A is partial pressure of A x is mole fraction of B in liquld phase in the mixture of two liquids A and B and y is the mole fraction of A in vapour phase then P B in mm of Hg
[desc-10] [desc-11]
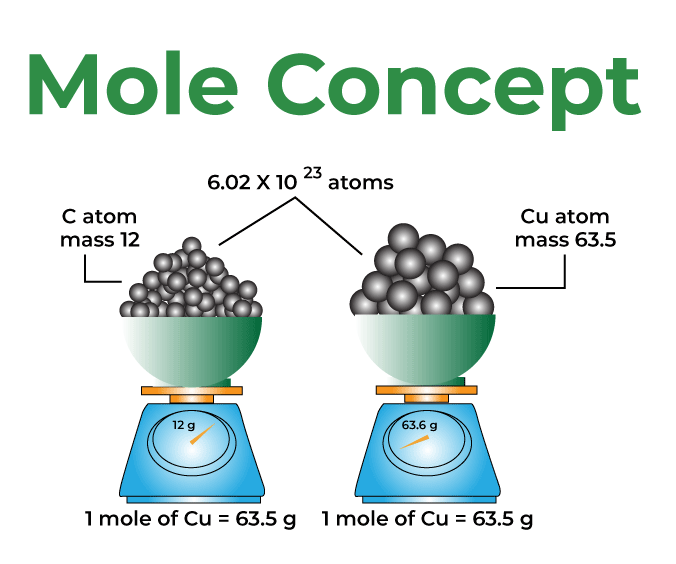
Mole Concept Definition Formula Examples And FAQs 59 OFF
https://media.geeksforgeeks.org/wp-content/uploads/20220923161052/Moleconcept11.png
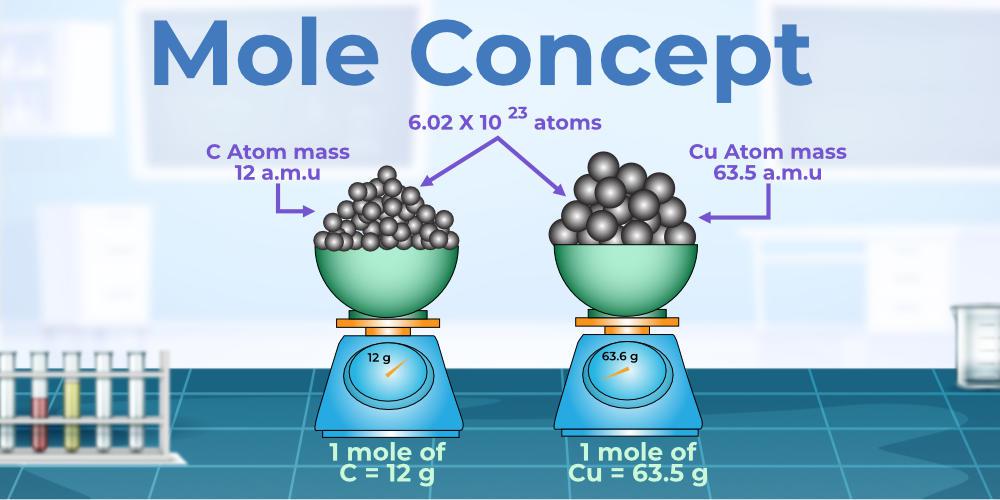
What Is A Mole Ratio Chemistry Definition And Example 51 OFF
https://media.geeksforgeeks.org/wp-content/uploads/20230306120352/Mole-Concept.jpg
Mole Ratio To Mass Ratio - One mole of an ideal monoatomic gas initially at 1200 K and 64 atm is expanded to a final state at 300 K and 1 atm To achieve the above change a reversible path is constructed that involves an adiabatic