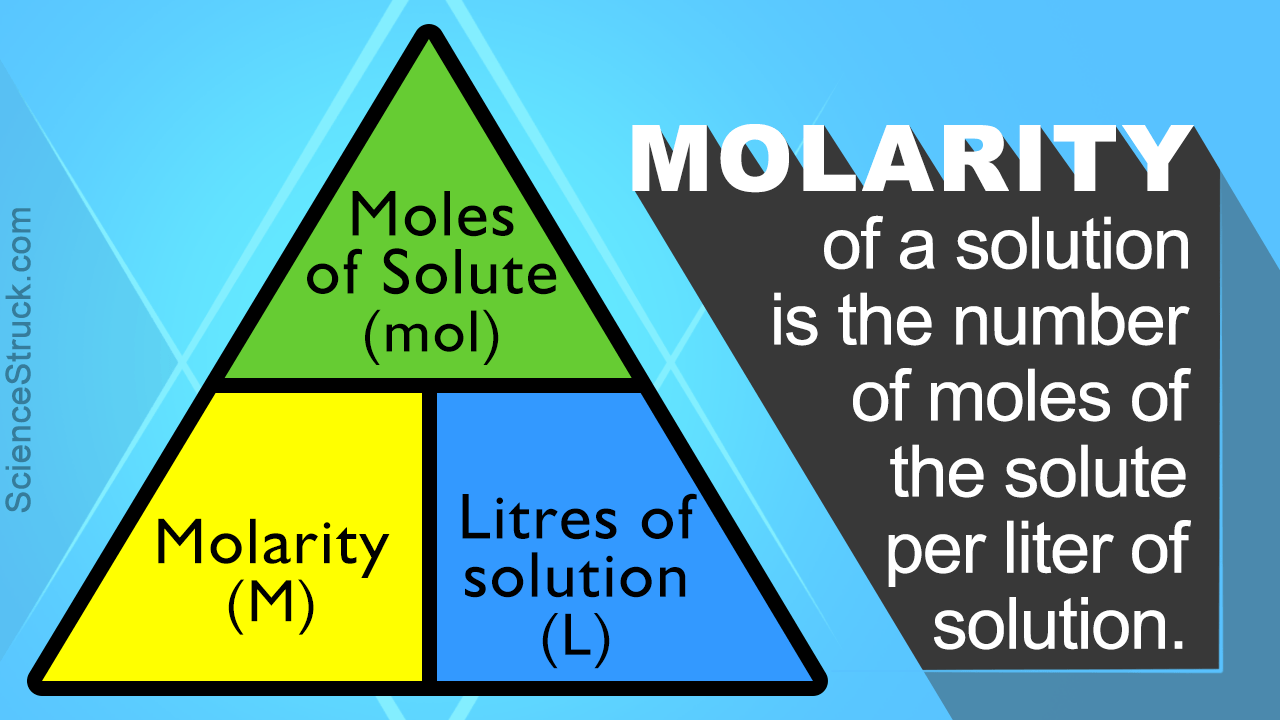
What Is Molality Molarity And Mole Fraction What is molarity What is molality Compare molarity vs molality by viewing molality and molarity formulas how to calculate molarity and molality and examples
Molality may be a measure of the concentration of a solute during a solution in terms of the quantity of substance during a specified amount of mass of the solvent Formation of term molality is in analogy 5 Molarity is affected by changes in pressure while molality is unaffected by changes in pressure 6 Molarity may result in an imprecise and inaccurate concentration while molality results in an accurate
What Is Molality Molarity And Mole Fraction

What Is Molality Molarity And Mole Fraction
https://classroom-images.cdn.askfilo.com/classroom/1675432974115_xvtjhpgn_1923692.jpg
Molarity Molality And Mole Fraction Exercises PDF
https://imgv2-1-f.scribdassets.com/img/document/905699599/original/625ac8091c/1?v=1
Molarity Molality Mole Fraction Quiz
https://quizizz.com/media/resource/gs/quizizz-media/quizzes/de69fbbb-9a8c-4c6d-9971-625544a284b2
Molality is defined as the number of moles of solute present per 1000g of solvent So the molarity is function of volume which can be affected by changing the temperature of system while the molality of About This Quiz amp Worksheet This quiz and corresponding worksheet will help you gauge your understanding of how to calculate molarity and molality concentration
Molality It is defined as the moles of the solute present in 1 kg of the solvent It is denoted by m Molality is preferred over Molarity then why for determining molecular maas we prefer osmotic pressure method it also uses molarity
More picture related to What Is Molality Molarity And Mole Fraction

Molarity And Mole Fraction Definition Formula Questions And Examples
https://cache.careers360.mobi/media/article_images/2025/9/1/Molarity And Mole Fraction.jpg

PPT Molality And Mole Fraction PowerPoint Presentation Free Download
https://image3.slideserve.com/6594493/molality-and-mole-fraction-n.jpg

Molality Equation
https://pbs.twimg.com/media/FLzvfECakAMitpL.jpg
Molality m of a solution is defined as the number of moles of the solute present in one kilogram of the solvent Molarity is temperature dependent while molality is independent of the temperature If molality of the dilute solution is doubled the value of molal depression constant Kf remains unchanged
[desc-10] [desc-11]

PPT Molality And Mole Fraction PowerPoint Presentation Free Download
https://image3.slideserve.com/5494305/molality-and-mole-fraction-l.jpg

Molarity Molality Or Mole Fraction 3D Animated Explanation
https://i.ytimg.com/vi/zJLwgM0b_uA/maxresdefault.jpg
What Is Molality Molarity And Mole Fraction - [desc-12]