Mole To Mole Conversion Stoichiometry Atomic mass is the concept related to a single atom whereas molecular mass relates to a group of atoms If you are planning to proceed with this concept then it is necessary to gain precise
Mole and Equivalent Weight If we ask you to count the total number of stars in the sky can you do that No Similarly scientists can not count the exact number of atoms and molecules as they are very tiny A magnesium ribbon when burnt in air left an ash containing M gO and M g3N 2 The ash was found to consume 0 6 mole of HCl when it was taken in solution according to the following reaction M g3N 2
Mole To Mole Conversion Stoichiometry

Mole To Mole Conversion Stoichiometry
https://worksheets.clipart-library.com/images2/moles-to-mass-worksheet/moles-to-mass-worksheet-12.jpg

Moles Conversion Worksheet
https://i.pinimg.com/originals/f0/1d/04/f01d04a7a37c445e287cb5618ec8c79b.jpg

Unit Stoichiometry Answer Key Mole Mole Calculations WS 1
https://worksheets.clipart-library.com/images2/stoichiometry-worksheet-mole-mole-answers/stoichiometry-worksheet-mole-mole-answers-26.png
Vapour pressure of chloroform CH Cl3 and dichloromethane CH 2Cl2 at 298 K are 200 mm H g and 415 mm H g respectively i Calculate the vapour pressure of the solution prepared by mixing 25 5 g Assuming that 1 3 5 hexatrience has only pure double bounds and pure single bonds how many grams of it contain one mole of double bonds
If EC C is 344 kJ mole 1 and EC H is 415 kJ mole 1 calculate the heat of formation of propane The heats of atomization of carbon and hydrogen are 716 kJ mole 1 and 433 kJ mole 1 respectively An amount of 1 mole of N 2 and 4 moles of H 2 are allowed to react in a vessel and after reaction water is added Aqueous solution required 1 mole of H C l for complete reaction
More picture related to Mole To Mole Conversion Stoichiometry

Free Mole Conversion Worksheet Key Download Free Mole Conversion
https://worksheets.clipart-library.com/images2/mole-conversion-worksheet-key/mole-conversion-worksheet-key-36.png

Extended Reaction Stoichiometry Road Map Examples Expii
https://d20khd7ddkh5ls.cloudfront.net/img_5bd217010b9e-1.jpeg

18 Mole Conversion Problems Worksheet Answers Free PDF At Worksheeto
https://www.worksheeto.com/postpic/2010/07/mole-conversion-worksheet-answers_208035.png
Molal depression of freezing point of water is 1 860 per 1000 g of water 0 02 mole of urea dissolved in 100 g of water will produce a lowering of the freezing temperature by The henrys law constant for the solubility of N 2 gas in water at 298 K is 1 0 215 10 5 atm the mole fraction of N 2 in air is 0 8 the number of the moles of N 2 from air dissolved in 10 moles of water at 298 KL
[desc-10] [desc-11]

Stoichiometry Example 3 Mole Of Reactant To Mole Of Reactant
https://i.ytimg.com/vi/w-ty7HOzPLE/maxresdefault.jpg
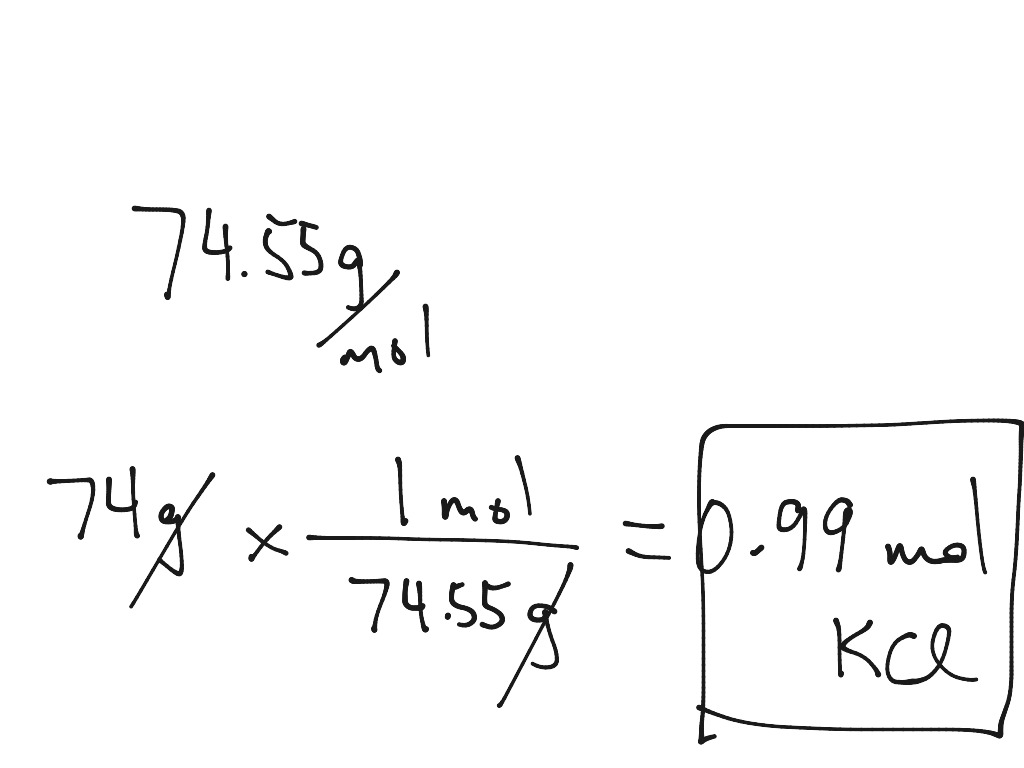
Gram To Mole Conversion Science Chemistry Stoichiometry ShowMe
https://showme0-9071.kxcdn.com/files/465871/pictures/thumbs/1895799/last_thumb1423583795.jpg
Mole To Mole Conversion Stoichiometry - If EC C is 344 kJ mole 1 and EC H is 415 kJ mole 1 calculate the heat of formation of propane The heats of atomization of carbon and hydrogen are 716 kJ mole 1 and 433 kJ mole 1 respectively