Mole To Mass Conversion Examples Mole standard unit 6 02214076 x 10 23 in chemistry for measuring large quantities of very small entities such as atoms molecules or other specified particles
What is a Mole In the field of chemistry a mole is defined as the amount of a substance that contains exactly 6 02214076 1023 elementary entities of the given substance The number Jul 10 2024 nbsp 0183 32 If you take chemistry you need to know about moles Find out what a mole is and why this unit of measurement is used in chemistry
Mole To Mass Conversion Examples

Mole To Mass Conversion Examples
https://martinlindelof.com/wp-content/uploads/2023/11/mole-conversions-solutions-worksheet-exercises-chemistry-docsity_0.png

Stoichiometry Lessons TES Teaching Chemistry Chemistry Education
https://i.pinimg.com/originals/26/18/6c/26186c154c2319bc82195d11c6cea15d.png

Molar Mass Conversion Worksheet Worksheet Worksheets Library
https://worksheets.clipart-library.com/images2/mass-to-moles-worksheet/mass-to-moles-worksheet-17.jpg
Jun 25 2014 nbsp 0183 32 A mole is a SI base unit of measurement This is a definition of unit its value and example problems using the mole in chemistry Dec 1 2023 nbsp 0183 32 A mole is a unit of measurement that is used to express the amount of substance in a chemical reaction The term mole is defined as the quantity or mass of a substance that
SI base unit mole mol The mole symbol mol is the SI unit of amount of substance One mole contains exactly 6 022 140 76 x 10 23 elementary entities This number is the fixed numerical Sep 21 2022 nbsp 0183 32 The mole is the unit of measurement in the International System of Units SI for amount of substance It is defined as the amount of a chemical substance that contains as
More picture related to Mole To Mass Conversion Examples
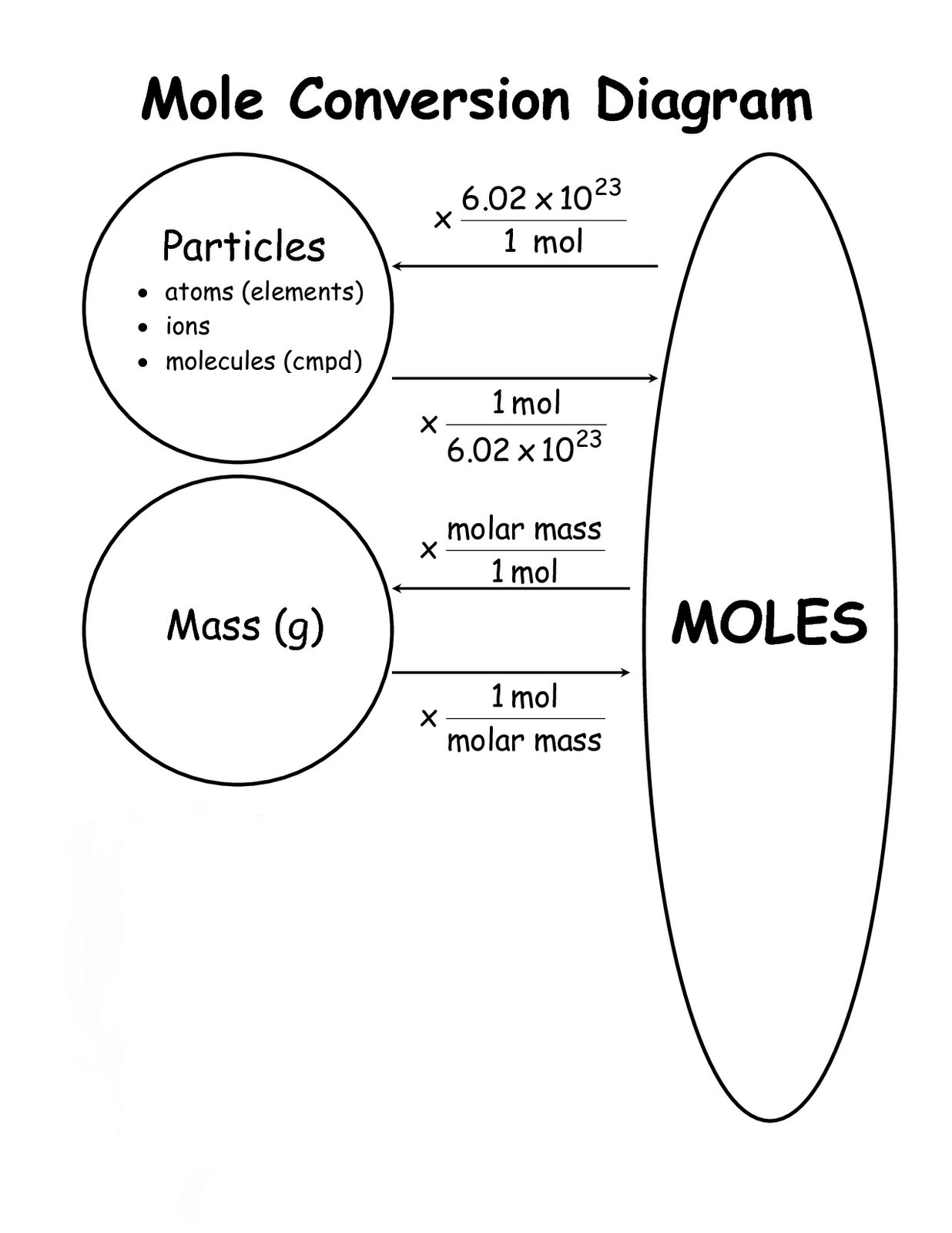
Converting Moles Worksheets
https://2.bp.blogspot.com/-9pWvVHv8bLM/TscWi7-jw1I/AAAAAAAAAIQ/cpJ0jZvKLks/s1600/16826138.png

Molecular Hydrocarbons Mole Conversions Vrogue co
https://image1.slideserve.com/2012278/mole-conversion-examples3-l.jpg

Mole Conversion Practice
https://i.ytimg.com/vi/3zmeVamEsWI/maxresdefault.jpg
Definition of Mole What is a Mole A mole corresponds to the mass of a substance that contains 6 023 x 10 23 particles of the substance The mole is the SI unit for the amount of a substance Mar 3 2025 nbsp 0183 32 The mass of one mole atoms of any element is exactly equal to the atomic mass in grams gram atomic mass of that element For example the atomic mass of aluminum is 27u
[desc-10] [desc-11]

Moles To Mass Conversion Worksheet Free Worksheets Printable
https://i0.wp.com/s3.studylib.net/store/data/008990530_1-bd019adc405d66f32c0cd09b0fbd143d.png

Mole Mass Conversion Practice
https://i.pinimg.com/originals/2d/79/6f/2d796f9c2845de3c49f3d21c3720cb16.png
Mole To Mass Conversion Examples - Sep 21 2022 nbsp 0183 32 The mole is the unit of measurement in the International System of Units SI for amount of substance It is defined as the amount of a chemical substance that contains as